In This Issue
Gait changes mark early neurodegenerative disease
Skin sensor detects organ failure
Slowly closing in on causes for post-acute infection syndrome
Survivors of childhood cancers are under-screened
New and Noteworthy
To diagnose early neuro disease, “throw ‘em a curve”
We’ve focused a lot lately on the explosion of biomarker tests for early diagnosis of neurodegenerative diseases such as Alzheimer’s and Parkinson’s. A small new study shows that a non-invasive, observational test may also be able to diagnose neurodegenerative disease in its earlier stages. Just watch the patient walk - on a curved path.
Previous research (2018) had shown that Alzheimer’s patients walk significantly more slowly than healthy folks do, especially on a curved path. This study looked at 50 different gait markers and found that 13 of them differed significantly between people with mild cognitive impairment (which can be an Alzheimer’s precursor) and healthy folks of roughly the same age. As in the 2018 research, larger differences were seen when study subjects walked a curved path as compared to a straight one.
Non-invasive colorectal cancer diagnostic options expand
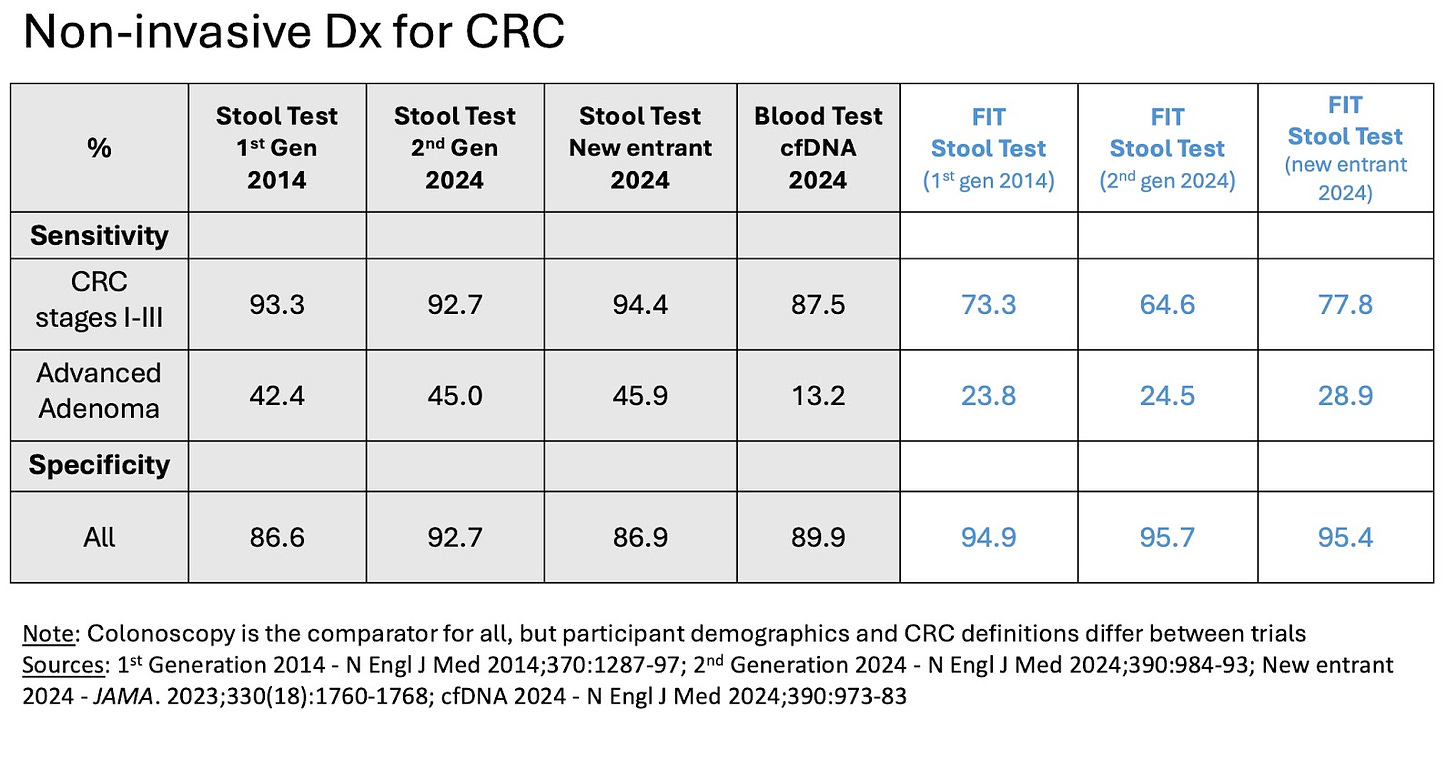
It’s been a big week for early detection of colorectal cancer (CRC). The New England Journal of Medicine published trial results for both a second generation stool-based test and a novel cell-free DNA (cfDNA) blood-based test, together with a brief summary of CRC cfDNA science and an editorial advocating for increased screening.
The addition of these two tests allows a comparison of four non-colonoscopy options for CRC diagnosis (we profiled another stool-based test in our February 15 newsletter, and the first-generation stool-based test paper appeared in 2014). Colonoscopy’s big advantage remains the fact that it enables the clinician to remove any precancerous adenomas (aka polyps or advanced adenomas) at the moment when they are diagnosed. Its big disadvantage is the time and effort that the procedure takes for the patient.
Commentary: A true apples-to-apples comparison of the research behind each non-colonoscopy test isn’t quite possible. However, we can at least say that all of them are better than the fecal immunochemical test (FIT). A FIT test simply looks for blood in the stool. The three newer non-colonoscopy tests were each twice as good as FIT at finding early adenomas, with one-third the number of false negatives. The cfDNA (circulating free DNA) blood test is reasonably sensitive at detecting stage I to III CRC - just a little less than the stool tests, but not as good at detecting adenomas as either FIT or stool-based tests.
Increased screening is essential because CRC incidence is growing, especially in younger cohorts (those 45 and older). Colonoscopy alone is unlikely to close the unscreened gap - it is just too expensive and time-consuming - so these tests serve a very important role.
Stool-based test sample collection can be done at home. It’s a little icky, but really no worse than changing a diaper. For those who have regular access to a physician, blood-based tests can easily be added to an office visit lab order. Growth in both sample types is most likely.
Skin patch that uses radar to sense organ changes
This week we were fascinated by the potential of a new type of diagnostic, a small skin sensor. Developed at MIT, the sensor is a sticker the size of a postage stamp. It acts like a radar system, “pinging” internal organs with sound waves and using the reflected pings to detect changes in the organs’ size or composition.
To date, the sensor has shown that it can detect liver failure in rats (when the liver fails, it gets rigid). If it works in humans, we’ll be able to get early signals (pun intended) telling us whether a treatment or disease is causing organ damage.
Alzheimer’s test no longer DTC
Remember that direct-to-consumer (DTC) Alzheimer’s test we reported on back in August and December? This week, MedPage Today reported that it’s no longer available on the company’s consumer platform, following “multiple communications involving Quest, the Alzheimer's Association, and biomarker researchers.” The test has not been approved by the FDA and has only been marketed as a non-diagnostic laboratory-developed test. It is still available by prescription.
Food for Thought
Diagnosing post-acute infection syndrome - light at the end of the tunnel?
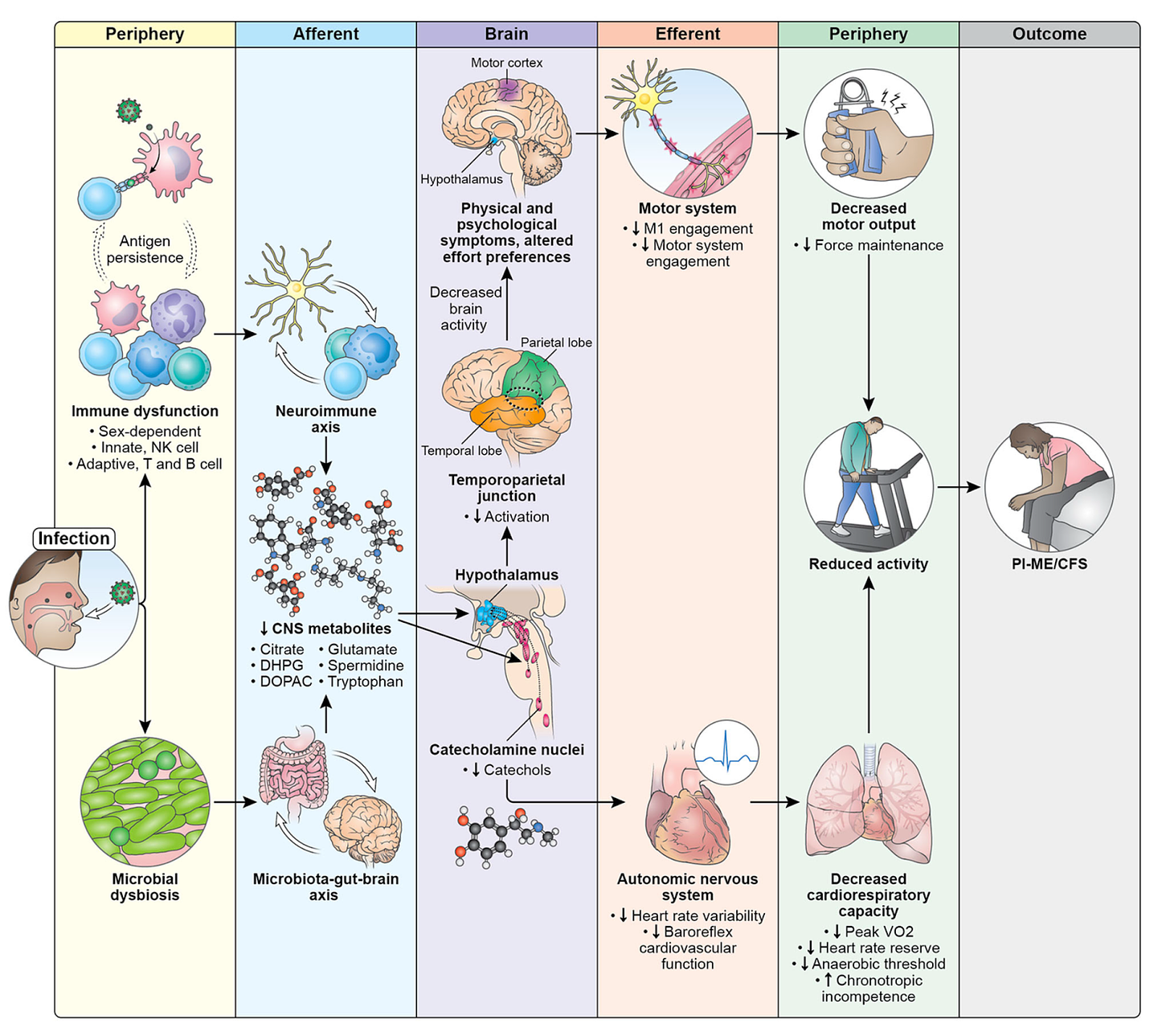
Long COVID is one of the lingering dark clouds of the pandemic. Its only silver lining is the focus it has brought to post-acute infection syndrome (PAIS) - the broad range of issues that can persist after many infections. Typically, patients with this condition have received the (somewhat abstract) diagnosis of myalgic encephalomyelitis / chronic fatigue syndrome (ME/CFS). But with neither a reliable, objective biomarker nor any effective treatments, the condition has been clinically neglected. Last month Nature published a comprehensive ME/CFS analysis (but based on only 17 patients), concluding that:
These patients suffer real and widespread physiological dysfunction (see Figure 10 extract below)
An overactive but ineffective immune response is a common driver
Specific B- and T-cell biomarkers distinguish ME/CFS from healthy controls
Commentary: Chronic Fatigue Syndrome, Long COVID, and similar conditions are real. Too many people, especially women, have been told that their symptoms exist only in their imagination and are not worthy of medical intervention. We hope and expect with this and related studies that more will be done to focus on the underlying causes and treatment for these conditions.
Childhood cancer survivors need follow-up testing - for life
Cancer is the second-highest cause of death of children (17.3% of childhood deaths). When treatment allows a child with cancer to go into remission and survive into adulthood, it is an event for celebration. But that win comes at a high cost. Up to 80% of these survivors will experience severe or potentially fatal health issues as a result of the treatment that cured their cancer. Childhood cancer survivors as a group have a risk of heart disease or breast or colorectal cancer that is two to three times higher than that of the general population.
Why is this relevant to diagnostics? A large study just published in the Canadian Medical Association Journal showed that, for reasons that are not clear, childhood cancer survivors have a lower-than-average diagnostic screening rate - even when they’re treated at academic centers. The percentage of these folks who meet industry screening guidelines is alarmingly low: only 6% for breast, 13% for colon and 53% for cardiomyopathy.
Commentary: Today’s adult survivors of childhood cancer were treated at a time when it was assumed that more treatment was better. We believe (hope) that children treated today will suffer fewer (but not zero) long-term negative effects from their treatment. But even if we are right, that does not change the need for better adherence to testing guidelines right now.
Quick Hits
Patients who have undergone surgery on their gastrointestinal tracts must be closely monitored for leaks during their first week post-op. While such complications can be fatal, they’re tough to detect. A new implanted, bioresorbable device could make detection easier. If the abdominal pH changes due to a leak, the device’s shape changes in a way that can be measured with ultrasound.
Only 20 to 25 percent of cancer patients respond to immunotherapy, a treatment that uses the patient’s immune system as a weapon against cancerous cells. A small study of 60 subjects found a set of 140 genes whose presence in patients with non small-cell lung cancer may indicate that those patients will respond well to immunotherapy. Patients with that genetic profile had “enhanced disease-free survival” when treated with a combination of an immunotherapeutic drug and low-dose radiation, as compared to those without.