In This Issue
Alzheimer’s blood tests must work IRL
In a molecular age, should we still be naming cancer by tissue?
At-home stool sample collection vs. colonoscopy
Five-day COVID isolation might end this spring
New & Noteworthy
Alzheimer’s disease blood tests face the real world
To date, trials that have tested the accuracy of bloodborne biomarkers for Alzheimer’s disease have generally targeted specific patient groups in which performance is expected to be strong (e.g., patients with specific cognitive score levels and with minimal co-morbidities). A recent Alzforum reports upcoming clinical trials (SUNBIRD, US; REAL AD, Sweden) to test the newest and best protein biomarkers in blood (Aβi 42/40, pTau217, etc.) in real-life primary-care settings. Some additional possibilities for the future: A recent large-scale study in Nature Aging that looked at blood samples in healthy adults identified four proteins (GFAP, NEFL, GDF15 and LTBP2) that, when present at high levels, were associated with the development of dementia over the next 15 years.
Commentary: In real life, patients are more diverse - from the worried well to those in ongoing cognitive decline. When used as a screening tool in primary care, lower prevalence threatens to generate false positives that could overwhelm accuracy.
Inexpensive and less invasive tests are essential to the identification of the patients and the related development of therapies that can be used earlier in the progression of disease. If Primary care will be in the hot seat: That’s where they must be used if they are to be effective.
Colorectal cancer screening alternatives to colonoscopy
The standard of care for the diagnosis of colorectal cancer (CRC) is still the invasive, time-consuming, and expensive colonoscopy. While its sensitivity is high for active disease (94.4%), it’s only 45.9% for the earliest forms of cancer (advanced adenomas). Plus, the test has high rates of non-compliance, especially among younger age groups - where its incidence has been growing.
In theory, a solid home test for CRC could improve screening rates (though anything that tests for this disease is likely to run rump-first into a squeamishness barrier). One such test with significant volume is already on the market. A second one was validated through the CRC-PREVENT trial, the results of which appeared in a recent JAMA paper.
Both tests require the user to gather a larger fecal sample for genetic testing and a separate, smaller sample for fecal blood testing. The older of the two tests detects DNA, while the newer one looks for RNA in an attempt to reduce false positives due to age-related DNA methylation patterns. The CRC-PREVENT trial did not directly determine whether RNA is in fact a better target, other than to comment that among the subgroup with a previously negative stool test, advanced adenoma sensitivity was higher at 61.7% (but there are many other possible reasons for this). FDA 510k approval (510K) is pending.
Is five-day COVID isolation on the way out?
One of the last pandemic-era restrictions that remains in everyday life may not be around much longer. This week, the Washington Post announced that the CDC was planning to get rid of the mandatory five-day isolation guideline for folks with COVID. Instead, they reported that the agency was going to switch to a symptom-based system that mirrors recommendations for people sick with flu. The change would reportedly take effect sometime this coming April.
When pressed for confirmation, CDC officials gave the public-health version of “no comment,” saying only that “We will continue to make decisions based on the best evidence and science to keep communities healthy and safe.”
Commentary: We get it. Isolation sucks. And it’s a disincentive to testing, because people don’t want to test positive and then have to isolate especially if you don’t have many or any symptoms. But the bottom line is that COVID, however mild, is highly contagious. You and your family may all be relatively young and healthy, but others are more vulnerable - or their loved ones may be. And this goes for diseases beyond COVID. We would feel differently about potentially eliminating isolation if masks were used broadly. But - alas - they are not.
Food for Thought
Personalized medicine goes back to the future?
Cancer’s molecular revolution of the past 25 years has been fueled by a growing portfolio of drugs that target (molecularly diagnosed) driver mutations one by one. These mutations typically involve overexpression of an oncogene such as HER2 or loss of a tumor-suppressing gene such as p53; a good example is Imatinib/Gleevec, which has a track record of 98% remission at five years for previously fatal CML.
The search for such precisely targeted treatments has expanded past oncology into other diseases, past single-gene mutations to those involving multiple genes, past genetics to other diagnostic modalities, and past drugs to other treatments. And it continues to log incremental successes, in its painstaking, one-study-at-a-time way. Some recent examples include:
An AI-based study showing that hydroxyprogesterone caproate injection helps prevent preterm birth in patients with a certain genetic signature
The discovery that tumors with a higher intragenic rearrangement burden (IRB - those with more mutations involving big chunks of genetic code rather than mutations involving single base pairs) may respond better to immunotherapy than those with lower IRB
Confirmation that biodynamic imaging (using doppler ultrasound to measure whether the motions inside cancer cells change after exposure to a chemotherapeutic drug) can tell whether a given chemo drug is working in a particular patient
The development of a machine-learning model that can tell whether lumbar disc herniation surgery is likely to work well in a given patient
This approach has thus far helped many people. However, in June 2023, the NCI-MATCH clinical trial designed to evaluate molecularly driven drug treatment across a range of cancers reported disappointing results – improvements in some cases, but an average response rate of only 10.3% overall.
A feature in this week’s Nature described an entirely different route to precision medicine in oncology: Just take cells from the patient and treat them with everything available that might work. When you find something that works in-vitro, give it to the patient. At least one trial is underway to see whether this approach, called functional drug testing, might be more effective than the genome-directed standard of care.
Commentary: Functional drug testing may be an effective approach, but important to note that it is not new. Many companies have tried matching drugs to patient tumor tissue in vitro but were not able to execute at scale or show efficacy in multiple tissue types. But, before the molecular age, virtually all cancer diagnostics were tissue-based. Molecular diagnostics have not obsoleted tissue diagnostics - the new paradigm may be pan-diagnostics, requiring a more integrated holistic patient assessment of molecular dynamics within cells within tissues within organs.
Is it time to ditch a 2000-year-old cancer paradigm?
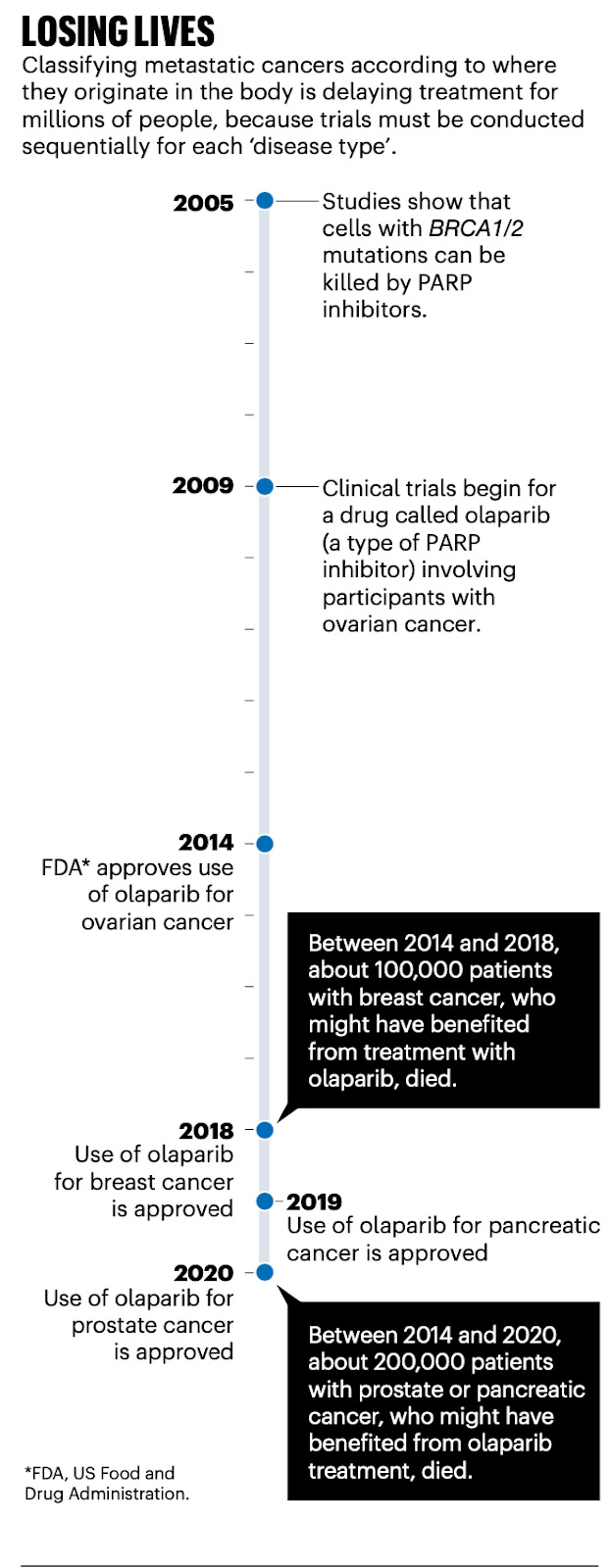
Since the birth of the genomic age 20 years ago, there has been ongoing discussion about whether it is now time to use genomic (aka molecular) cancer descriptions instead of the tissue-of-origin paradigm established in the days of the Roman Empire (Galen). This week, Nature published an interesting commentary from French oncologists titled “The way we name cancers needs to change.”
On the pro-change side: Molecular diagnostic descriptions are more clinically relevant to gene-mutation-targeted drug treatment. Included in the commentary is an example of how long it took to bring one particularly effective drug (Olaparib for ovarian cancer, reproduced here) to other likely targets. On the anti-change side sit surgeons and interventional radiologists, for whom tumor location is an integral part of both diagnosis and treatment.
Commentary: This doesn’t have to be an either/or choice - almost all cancer patients’ treatments comprise a mix of at least two of these three approaches. Additionally, most cancers have both molecular and tissue-specific dysfunctions. Just because a drug is effective in one tissue does not always mean that the same drug will extend overall survival across other tissues. Even Olaparib has differential survival from tissue to tissue.
Quick Hits
The FDA has approved the first smartwatch-based application to detect obstructive sleep apnea. It’s intended to diagnose “moderate to severe” forms of the condition in people age 22 and up.
We discussed diagnosis errors 2 weeks ago, based on newly released (pretty horrific) JAMA data on where in the patient’s pathway of care errors occurred for the most seriously ill patients. One steady advocate for the need and utility of AI done right in clinical care is Eric Topol, who recently published an update to a review he had written in Science - both are worth a read.