Also In This Issue
A new possibility for early pancreatic cancer dx
Liquid biopsy predicts (lack of) bladder cancer recurrence
Can gut microbiome indicate Tx-resistant schizophrenia?
Digital diabetes management underwhelms
The controversy behind the opioid-use disorder test
New and Noteworthy
New hope for detecting early stage pancreatic cancer
Pancreatic cancer is often a silent killer - by the time symptoms appear it is typically too late for successful surgery or drug therapy. This week, a novel approach to its early diagnosis was presented at the annual meeting of the American Association for Cancer Research (AACR): isolating and analyzing microRNA contained in circulating extracellular vesicles (EVs).

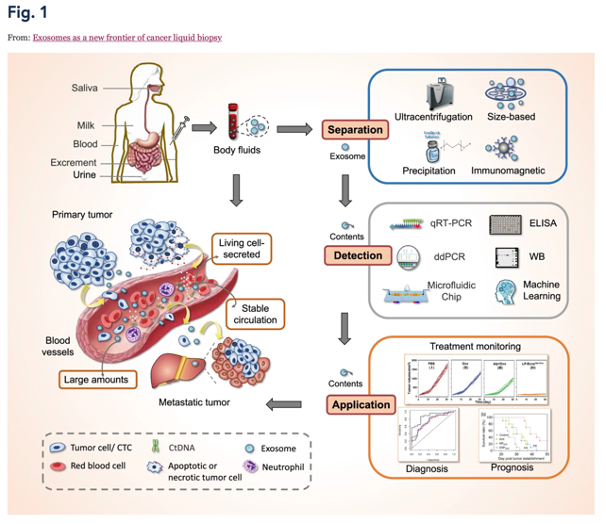
EVs are an especially promising target because they are released by all cells, including localized cancer cells (see this review, this February 2024 paper, and the graphic below from the Goel Lab; a more effective way to isolate them was recently reported, too). By contrast, circulating tumor DNA (ctDNA) is thought to be released later in tumor progression, only as cells die, which may help explain why liquid biopsies fail to detect the earliest stages of many cancers. In the test announced at AACR, 97% of early (localized stage I and II) pancreatic cancers were detected by combining a traditional cancer marker (CA19-9) with the EV panel.
Commentary: This work demonstrates that an effective early warning test for pancreatic cancer is potentially within reach, though not quite clinic-ready. It also demonstrates how much deep understanding of tissue-specific dynamics and microenvironments is required to develop accurate diagnostic tests.
Predicting (lack of) bladder cancer recurrence with liquid biopsy
Bladder cancer has the one of highest recurrence rates of all cancers: Nearly 75% of patients will experience recurrence within 10 years. So predicting those who will not recur would have huge value.
For this particular malignancy (unlike pancreatic cancer), a test for circulating tumor DNA (ctDNA) was highly effective. According to results of a trial presented at the 2024 European Association of Urology Congress, more than 90% of patients with a negative ctDNA test post-surgery did not relapse. These patients could potentially be managed much more conservatively than other bladder cancer patients (with less intense chemo, or no chemo at all). Interestingly, this diagnostic data was discovered as part of a therapeutic clinical trial looking at the efficacy of immunotherapy drug Tecentriq.
Could gut microbiome warn of drug-resistant schizophrenia?
The more we understand the role of our gut microbiome - the small universe of microbes we host in our GI tracts - the more important it becomes. The brain and gut are intimately connected (via the vagus nerve), and the gut microbiome plays an important role in curating communication between the two.
Many studies (see here for a review) have identified correlations between specific microbiome compositions and mental health. But for the most part there is no consensus on the clinical implications of microbiome differences between healthy individuals, nor of microbiome changes in the same individuals over time – a chicken-and-egg problem. Now a recent paper describes a possible way to use microbiome characterization to identify those who suffer treatment-resistant schizophrenia.
Commentary: Unfortunately the differences are too varied to support a viable diagnostic test. Plus it’s a small-ish study (n=97 split into four groups), and quite nuanced: Microbiome composition was found to be only marginally disrupted between healthy controls and successfully treated schizophrenic patients. Non-responsive patients, however, had less diverse and quite different microbiomes than controls.
Illustrating the lack of consensus, these conclusions tend to contradict prior papers that claimed these same microbiome differences were associated with a schizophrenia diagnosis alone. Either way, it is unclear whether treatment generates microbiome differences, or whether the microbiome causes the resistance. Only in the latter case can this be used to identify resistance in advance.
In genomic test development, diversity matters
Back in January, we mentioned that the FDA had approved the first genetic test to diagnose possible increased likelihood of developing opioid-use disorder (OUD). Turns out that approval was an unpleasant surprise to many in the genomics field. A group of about 30 experts from a wide array of academic and private institutions have sent letters to FDA and CMS asking them to withdraw / decline to fund the test.
The underlying issue was explained in a 2021 paper (cited in the letter). It evaluated a test that looks for the same set of genetic markers that the recently approved test looks for - and if you’ve been following our genomics coverage, the problem will sound really familiar. The algorithm behind the test doesn’t account for ethnic and racial diversity. According to the paper - and the expert letter - if you use this test on a group of people whose ancestry is diverse, it won’t predict risk of OUD any better than chance.
Food for Thought
Digital diabetes management solutions underwhelm
A recent report from the Peterson Health Technology Institute examines the effectiveness of eight digital diabetes-management solutions, all of which attempt to integrate a variety of clinically meaningful information into a simple mobile app. Six of the eight manage lifestyle and behavioral modification; one focuses on remote patient monitoring for patients and physicians; and one guides patients through nutritional ketosis (a diet based on protein and plant fats and oils, with very few carbohydrates).
However, while these programs accept device data from available meters, proprietary patient tools from the major makers of continuous glucose monitors were NOT included in this review. The report concluded that the ketosis diet-management tool was the only one that was successful and economically favorable.
Commentary: These apps could be very useful for those committed to major change, because we know that type 2 diabetes is reversible and avoidable. However, the majority of patients with type 2 diabetes and pre-diabetes are not so committed. Given that this was a meta-study of existing research not designed for this specific purpose, the relative success of the keto diet may be due not only to the app and the effective (but onerous) regime it prescribes, but to the fact that only the really committed are willing to sign up for such a diet.
Quick Hits
The International Diabetes Foundation has published a position statement advising clinicians to use a one-hour 75g oral glucose tolerance test (OGTT) to diagnose people at risk of developing type 2 diabetes. “Over the last 40 years, a wealth of epidemiological data has confirmed the superior value” of that test over the hemoglobin A1c test, fasting plasma glucose level, and the two-hour OGTT, the group stated.
What can’t smartphones do? According to recent studies, they can diagnose everything from ear infections (with sensitivity of 93.8% and specificity of 93.5%) to frontotemporal dementia (with better accuracy in early stages of disease than a standard cognitive assessment tool). And you said the tricorders on Star Trek were only science fiction.






Very true about the utility of liquid biopsy in tracking disease recurrence after treatment. I write about Grail, a commercially available liquid biopsy product for $850