We could sequence all newborns. But should we?
Volume 7, Issue 8 | February 22, 2023
In This Issue
Rapid antigen test sensitivity for Omicron
Marburg virus in Equatorial Guinea
Variant-wise, we got lucky in China
New and Noteworthy
Rapid antigen tests still work on Omicron - but timing matters
We have to be vigilant to ensure that the COVID test portfolio maintains its effectiveness as the virus evolves. The Omicron variant has nine N-protein changes as compared to the Delta variant, which is a concern given that antigen tests (which detect viral proteins) and rtPCR tests (which detect viral RNA) both focus on sub-regions of that protein. However, a recent preprint concludes that (at least through BA.1) eight commercial tests all retained sufficient sensitivity to detect both variants.
The authors found that both variants were equally infectious in the lab, but that on average, Omicron appeared to shed ~100 times less antigen for any given amount of mRNA than Delta did. Why should that be?
After examining and eliminating several other options, the authors infer that this result is really a reflection of when samples were taken. Omicron patients’ immune systems, primed by vaccination or previous infection, could have caused them to become symptomatic - and thus to be tested - earlier in the course of infection than the Delta patients. If that is indeed the case, it would explain the discrepancy: N protein levels don’t peak until a few days after RNA viral loads do. This difference in timing - and thus in levels of N protein - would also explain antigen tests’ reported decreases in sensitivity to the Omicron variant.
Marburg virus outbreak in Equatorial Guinea
The World Health Organization has confirmed that Equatorial Guinea is experiencing an outbreak of Marburg virus, a hemorrhagic-fever virus related to Ebola. Nine deaths have been recorded to date, and 16 additional cases are suspected. The WHO reports the fatality rate at 50% (previous outbreaks have seen deaths in close to 90% of those who are infected and do not get treatment, which consists solely of rehydration). WHO is hoping to test a vaccine for the virus during this outbreak.
Marburg is thought to have originated in African fruit bats. While it is not airborne, it can be contracted from person to person through direct contact with bodily fluids or through prolonged contact with contaminated surfaces.
Food for Thought
Is it time to begin universal newborn genetic sequencing?
Routine screening of newborns for congenital diseases started in this country in the 1960s, when dried blood spots were first obtained for all babies born in the US. Today, the federal Health Resources and Services Administration (HRSA) recommends newborn screening for 63 disorders - as with vaccinations, it is then up to the states to decide which of these recommendations to accept. According to the NIH, all states currently require newborn screening for at least 29 health conditions. While they allow an opt-out for religious reasons, participation is 99%+ today for the nearly four million babies born in the US.
Traditional newborn screening uses routine lab tests enabling physicians to screen, and then diagnose and treat 12,500 US babies born each year with a disease that would have been devastating if left undetected. (If a newborn screen comes back positive for an individual disease, the baby typically then gets a confirmatory test before treatment.) This works well in the current regimens - but screens only see the 29 to 60+ disorders depending on the state.
But we can do much better - there are ~5000 genetic disorders affecting ~140 million children worldwide, all of which can be diagnosed from a single whole genome sequence (WGS)
Newborn sequencing can change lives. Right now, most people with rare genetic diseases are only diagnosed well after they become symptomatic - a recent US survey reported that it takes an average of 7.6 years to receive an accurate diagnosis. That’s precious time lost. Once symptoms appear, treatment to arrest disease progression is often futile or minimally effective at best.
So why aren’t we sequencing every newborn? Next generation sequencing has made it increasingly feasible to use those same dried blood spots to obtain a whole genome sequence for newborns. Both the cost and the amount of time required to do that sequencing have significantly declined.
But significant ethical and logistical questions remain. How much of this enormously powerful information should be stored? By whom, and for what purpose? Who will have the right to access these genomes? Can they be effectively protected from data leaks? How will research use be managed? Should the majority of mutations, whose contribution to overall risk is minor, be reported at all? Will relatives be informed of their possible carrier status? Does the child’s health insurance cost change? Who pays for proactive treatment? Can the data be used in the future by law enforcement?
And speaking of data - if we sequence everyone, an enormous amount of data will be produced, and realistically, not all of it can be stored. A single individual can generate up to 150 terabytes of raw sequence data (BAM file). If only mutations are stored, the number is still 52 gigabytes (VCF file). The 3.7 million US children born each year would require a minimum of two exabytes a year - today, the whole of Amazon Web Services stores is estimated to store about an exabyte (one with 18 zeros after it).
Commentary: The technology exists to implement newborn NGS in addition or to supplement newborn screening - but should we? This is a knotty and complex question, but we have to start to address it for the sake of the children and families who are affected. About half of affected kids will become symptomatic during childhood - the remainder will show symptoms later in life. Many will be admitted to the NICU/ICU, and among these children, ~30% will not reach their fifth birthday.
The reality is that the ball is already rolling - and as we’ve seen with other technologies, we need to start now if we want to control the direction in which it travels.
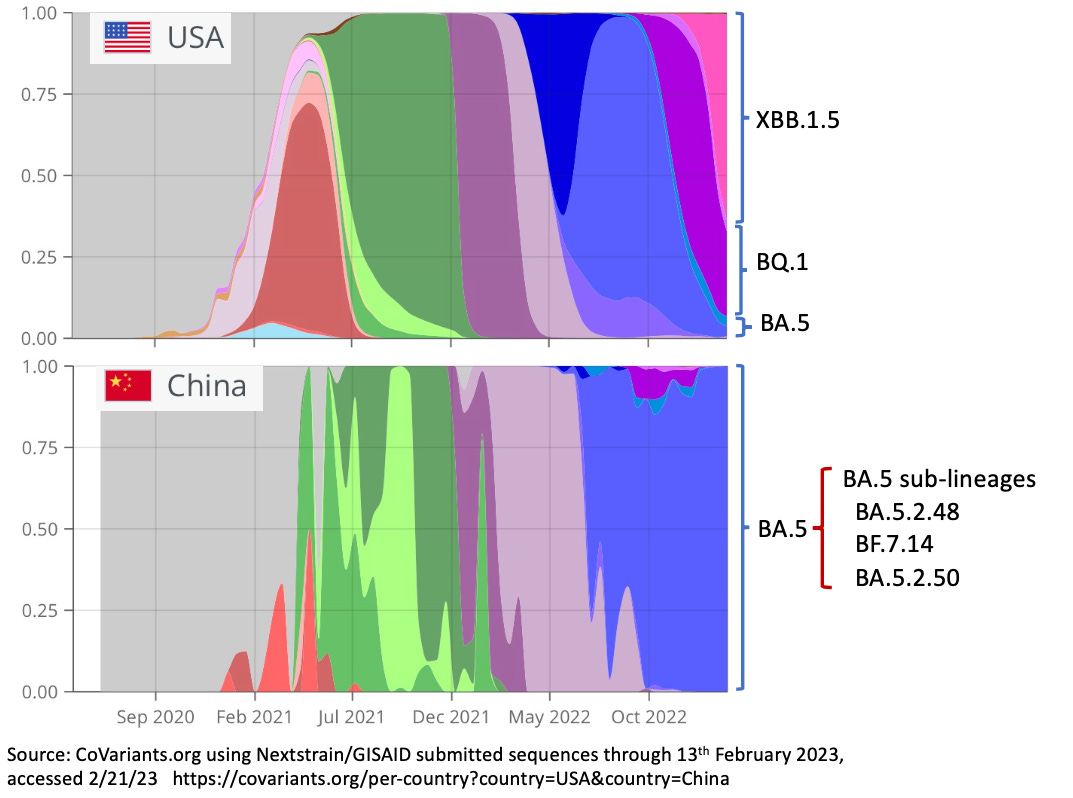
China played rock, paper, scissors with the global pandemic
As we discussed last week, China rolled the dice (apologies for the mixed metaphors) by abruptly ending their zero-COVID policy in December 2022. The world was concerned that novel variants with global consequences would result, but it looks like we have dodged the bullet - we have not seen anything very threatening yet, and cases in China are declining (although China is submitting very few sequences to international databases such as GISAID).
Which variant wins in a population is a function of pre-existing immunity and the winner’s ability to evade it (hence the rock-paper-scissors analogy), so each country’s variant trajectory is different. Due to strict lockdown, the Zero-COVID policy and relatively low vaccine uptake and effectiveness, especially among older citizens, China’s population was relatively immune-naive when the virus started blazing through at the end of last year. That meant the virus didn’t face significant pressure to evolve - it had plenty of easy-to-infect hosts available.
Commentary: It’s easy to say in hindsight that we didn’t need to worry - COVID has surprised us too many times for us to be comfortable in complacency. But it’s good to be able to breathe a sigh of relief for once.
Quick Hits
Moderna has announced that it will offer its COVID vaccine at no cost to people in the US who have no insurance. However, qualifying for it will require an application, a process which can be onerous for exactly the low-resource population the program is intended to help.