IN THIS ISSUE
Europe takes our advice on NGS in oncology ;)
State of the Tripledemic
End-of-year legislation and the diagnostics industry
Congress’s coronavirus lessons learned
Did you get sick after that in-person conference?
New and Noteworthy
NGS in Oncology Gets a Small Boost in Europe
Last week, we talked about the challenge of integrating next-generation sequencing (NGS) into oncology patients’ work-up. Seems like the EU was listening, as they just announced $5 million of grants to 11 institutions, for that exact purpose: integrating and standardizing NGS into clinical oncology.
Commentary: It’s a relatively small amount of money, but it will be great to get real-world evidence about the impact of NGS on cancer patients - and to give oncologists more experience with it.
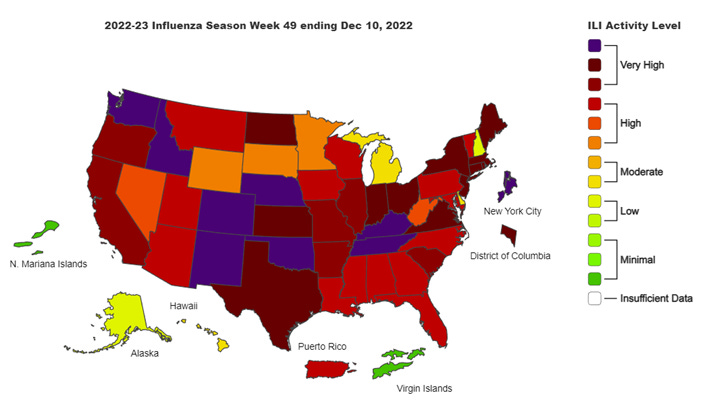
State of the Tripledemic: Still bad, especially for kids
The headlines say it all. In LA County, hospital beds are as full as they’ve been throughout the pandemic. In Philadelphia, schools are requiring masks again - to avoid the fate of certain districts in Indiana, Iowa, Maine, Michigan, Missouri, New Mexico, New York, and Texas, which have had to close due to illness. CVS and Walgreens are limiting purchases of children’s pain-relief medicine (which are also fever reducers), and manufacturers say supplies may not bounce back until spring. Tamiflu and amoxicillin are also in short supply.
While both RSV and flu levels have decreased a bit (see map below), the changes haven’t been enough to take the strain off of hospitals, especially in pediatrics. And there’s no guarantee that the levels will continue to go down - especially when it comes to flu. Commentary: Be careful out there - and remember to protect those who are most vulnerable, including the elderly, the newly born, the unvaccinated and those who are immunocompromised.
Congress’s “Lessons Learned” has some good stuff, once you get past the politics
The House of Representatives’ Select Subcommittee on the Coronavirus Crisis has published their final, 200+page report, Preparing for and Preventing the Next Public-Health Emergency: Lessons Learned from the Coronavirus Crisis.
Commentary #1: It’s far too focused on highlighting the politics for our taste. But that aside, we like the list of 30 recommendations for how to “mitigate the risk of the coronavirus” moving forward, including:
Maintaining at-home testing capacity and developing better tests for common respiratory viruses
Promoting ventilation and filtration systems
Increasing and sustaining investment in public health, including data systems and skilled staffing
Modernizing public health communications
Increasing collaboration with global partners with an eye to equity
Supporting equity at home with measures like universal paid sick leave, permanent investments in the child-care sector, and investment in housing affordability.
Commentary #2: We would add one more action to best protect us against another viral surge: Focus on utilizing the NGS technology that was massively expanded for COVID variant tracking so that it can be applied to infectious diseases surveillance and diagnosis more broadly. In the developed world, COVID has enabled dramatic reduction of the cost and time to carry out detailed pathogen identification: A recent Nature News item reported that the SARS-CoV-2 genome can now be sequenced for less than $20, compared to $1,000 at the start of the pandemic. If this capability is not maintained and used, we will have wasted the opportunity to respond rapidly to the inevitable next pandemic.
Putting Lab Leftovers to Use in National Respiratory Surveillance
An interesting new comprehensive national respiratory virus variant surveillance system may be getting started in the US. The program uses what seems to us to be a novel sample source: residual samples from patient testing at hospital labs. As a starting point, the CDC has awarded $5 million to Helix to identify and track over 30 respiratory viral variants (including SARS-CoV-2, flu, RSV, and more) in samples coming from two health-care systems. The samples will be linked to deidentified electronic health record (EHR) data, potentially allowing researchers to link variants with clinical symptoms. To make this data truly effective, they are planning to proactively integrate and communicate with local health system partners will be necessary.
Inside Baseball - Inside the Beltway
The end-of-year Congressional sessions always bring a bit of a rush of legislation. This year, the result for the diagnostics industry has been more positive than usual:
Reimbursement for specimen collection (money a diagnostic lab gets for taking your blood, etc.) will move from $3.00 to $8.57. While the increase might seem steep, this fee has not changed at all since 1984.
The Protecting Access to Medicare Act (PAMA), which had been set to go into effect in January, was postponed for one year. In general, this new Medicare reimbursement system would have lowered reimbursement rates for tests, so the delay is good news for the industry.
The VALID Act may not become valid after all, as it appears to have been cut from final legislation. Not enough time or space to go into the details here, but VALID intended to combine the two different diagnostic approval systems into one, all under the FDA.
Food for Thought
Don’t let that industry conference become a COVID incubator!
Commentary: We are starting with commentary because we love this idea. Conferences in every industry are mostly in-person events once again, especially in the medical and scientific communities. In this piece published in Nature, the author strongly recommends that conference organizers create a post-conference poll amongst attendees as to whether people contracted COVID (or other respiratory illness) 48 to 72 hours after the conference. In his example - with 80% response rate - 28% of attendees reported contracting COVID. In our perfect - unfortunately fantasy - world, everyone would test after attending a large event. Since that is not going to happen, the survey itself may be a good proxy. Asymptomatic cases will probably get missed, but we aren’t about to let the perfect be the enemy of the good.
Quick Hits
Screening people entering a hospital for COVID symptoms, exposure, or travel costs a lot and yields only marginal benefits, according to a large study done at Yale New Haven Hospital. The most bang for the buck was seen during the early stages of the pandemic (possibly because that was before people started fibbing about whether they had symptoms). One unexpected side benefit: Screening helped to counteract the pressure that health-care personnel feel to come to work even though they’re sick.
Rather than thinking of Omicron as “mild,” it’s probably more accurate to think of Delta as “more severe,” according to new research from Johns Hopkins University. For the unvaccinated patients the authors studied, Omicron was nearly as likely to cause severe illness and death as any pre-Delta strain was. Delta was more likely to result in poor outcomes than either Omicron or earlier strains. For vaccinated folks, the strain of SARS-CoV-2 they got infected did not make a significant difference.