OTC Test firsts: Continuous glucose monitor and COVID / flu antigen test
Volume 9, Issue 10 | March 7, 2024
In This Issue
Protecting the right heart while saving the left
Early Alzheimer’s diagnosis: Look at p-tau
PSA results highlight need for diversity in research
New and Noteworthy
First at-home COVID /flu antigen test gets EUA
As the flu season winds down, the FDA has granted the first emergency use authorization (EUA) for an at-home over-the-counter (OTC) antigen test for COVID, flu A, and flu B. Results for all three diseases are displayed in 10 minutes. (The agency previously authorized a LAMP-based 30 minute at-home COVID plus flu test.) This lateral-flow test has a similar protocol as other antigen-based tests: The label specifies taking the test twice over three days with a minimum interval of 48 hours between tests.
Commentary: We believe that this is a watershed moment for home testing, especially because this test uses technology that consumers are now highly familiar with. When COVID was at its peak, home tests were broadly used. But the question remains: Was COVID an exception or the start of a real home testing revolution? An OTC flu test, reasonably priced, may make the at-home testing shift possible.
Okay, yes, we know that this is an EUA, not full marketing approval. But we very much doubt that consumers will differentiate on that basis. We also don’t know the price. Since this test won’t be fully launched for at least a few weeks, the results (pun intended) will not be known until next flu season.
Continuous glucose monitoring goes OTC
This week, the FDA approved the first over-the-counter (OTC) device to continuously monitor blood sugar (glucose) in people who do not rely on insulin. This includes people with Type 2 diabetes as well as those with pre-diabetes or those just curious about their glucose levels. No prescription is required. (The device is not to be used for monitoring low blood sugar.)
Continuous glucose monitors (CGMs), invented 25 years ago, can revolutionize the lives of Type 1 diabetic patients, whose pancreases cannot produce insulin. CGM alarms protect against the severe risk of coma from low blood sugar, and continuous data enables tighter glycemic control to decrease risks of progressive kidney failure and blindness (see exhibit below).
There are about two million Type 1 diabetics in the US today. The population of Type 2 diabetics (folks who can produce insulin, but whose cells don’t respond to the hormone appropriately) is much larger, at 34 million in the US. Their disease is less severe day to day, but they share the long-term risks of sustained high blood glucose. It is this large and growing population that OTC devices target.
Commentary: There is no question that wearable devices like this are the future of diagnostics for many indications (see our previous discussion for cardiac applications). This is also consistent with the trend of consumers owning their own health without physician intervention. (The recent legislation allowing the sale of hearing aids without a prescription is another example.) Yet just how many will need an OTC version of a CGM is not clear. Plus, while prescription CGMs are typically covered by insurance, an OTC device probably won’t be, at least initially.
To spot pre-symptomatic Alzheimer’s, look at p-tau
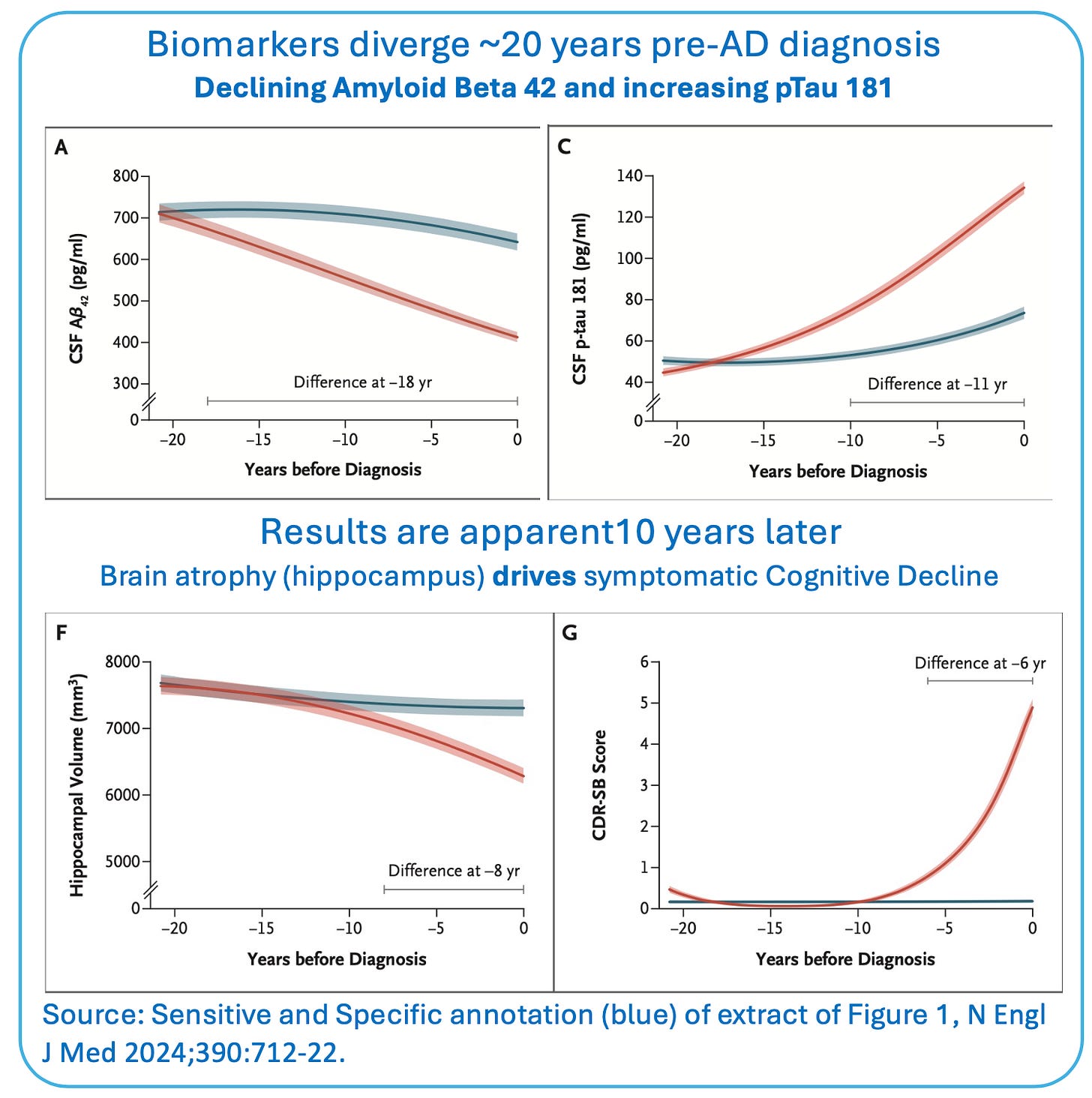
In January we reported on the value of phosphorylated tau 217 (p-tau217) blood tests to aid in the diagnosis of Alzheimer’s disease. Last month NEJM published the results of a 20-year longitudinal study to determine which biomarkers in cerebrospinal fluid (CSF) identify early Alzheimer’s. It confirms that changes in amyloid beta (Aβ) levels begin nearly 20 years before clinical diagnosis. Brain atrophy and cognitive symptoms can be identified 10 years after that, which is still 10 years before definitive diagnosis.
However, of the biomarkers evaluated, the winner was p-tau 181. Aβ42 levels change earlier than p-tau, but as shown in the figure below, they don’t as clearly distinguish those who go on to develop Alzheimer’s from those who don’t.
Commentary: Since we know that most plasma proteins are highly correlated with CSF proteins (less so for AB42), this paper is further strong support for the less-invasive blood p-tau tests now becoming available (plasma levels are generally about 5% of those in CSF).
High-quality data about diseases that take 20 years to reach diagnosis is almost impossibly challenging to get. This is the first longitudinal study (i.e., tracking the same patients over the years), and it took 32,061 participants in 2000 to end up with 648 diagnosed Alzheimer’s cases by the time the results were published. Nevertheless, there are issues. For one thing, testing technology and diagnostic criteria both evolve - blood tests for such low levels of protein did not exist when this study began.
One question this study leaves open: Why weren’t these patients definitively diagnosed until 10 years after imaging, biomarker, and cognitive tests all showed clear evidence of Alzheimer’s? The New York Times published an article this week that addresses this very issue. Effective treatments may be just over the horizon, but they aren’t here yet. Until we get them, perhaps cognitive symptoms have to become intrusive before a clinical diagnosis is useful.
Food for Thought
PSA provides a PSA about broadening diagnostic research
The vast majority of existing medical research was done using young adult Caucasian men as subjects. Increasingly, present-day researchers are discovering that results obtained only from that group may not be generalizable to other groups of people. A UK study recently found that Black patients had higher prostate-specific antigen (PSA) levels than their Caucasian counterparts, but fewer of them actually suffered from prostate cancer. The situation was reversed for Asian men.
Commentary: PSA level is already considered an unreliable biomarker. It is driven by prostate size, and an enlarged prostate is more often benign than malignant. These results provide yet more evidence that biomedical research has to focus on a broader range of potential patients and be careful to differentiate sub-group relevance.
Protecting the right side of the heart while saving the left
The left ventricle of the heart has a critical job: It pumps oxygenated blood through the body. When a person’s left ventricle suddenly stops working properly (as can happen due to a severe heart attack), doctors will typically put the patient on a mechanical pump called a left ventricular assist device (VAD). However, in 43% of patients, that extra assistance to the left ventricle can overload the right ventricle and make heart failure worse. The trick is knowing who that 43% is.
According to a recent study, there’s a way to diagnose that - and you can do it right at the point when you set up the VAD. You need to measure how stretchy the lung’s blood vessels are (a quality called vascular compliance). If those vessels are compliant enough, they’ll stretch to accommodate the extra blood that will come their way once the VAD is turned on. (The study established what “enough” means in terms of numbers.) If they aren’t, then blood will back up into the right side of the heart. The measurements can be made using the VAD itself along with data from a catheter into the main blood vessel feeding the lungs (routinely placed in patients like this).
Commentary: The study is small, with work done first in pigs and then validated retrospectively on data from only eight patients. But this is the kind of diagnostic - one that can be done just at the moment when it’s needed, with materials already available and familiar - that has the potential to immediately save lives.
Quick Hits
Is coffee bad for us? A new biomarker to help study this question
Google Scholar lists 2.8 million citations for studies on the effects that coffee has on our health. And though nearly all have failed to find any bad effects from normal levels of coffee drinking, study subjects may still feel a little sheepish about revealing how much they really drink. According to a paper published in Beverages, researchers can eliminate this self-reporting bias by using the N-methylpyridinium contained in roasted coffee beans as a biomarker. We can only hope that greater objectivity will not change the current consensus.
Are pre-surgical urine cultures necessary?
Even though current guidelines state that surgical patients only need pre-op urine cultures if they’re undergoing genito-urinary procedures, up to 25% of patients get cultured before other kinds of surgeries. Surgeons, listen up. A recent JAMA study confirms that pre-op urine cultures in non-genito-urinary cases don’t reduce infections - they just lead to delays and unnecessary antibiotic use. So knock it off.