Next-generation sequencing should be an oncology standard of care. Why isn’t it?
Volume 6, Issue 17 | December 14, 2022
IN THIS ISSUE
State of the Tripledemic
For some, it takes a while to get to Test to Treat
BQs KOs the last monoclonal antibody treatment
New and Noteworthy
State of the Tripledemic: Not Good
According to a CNN analysis, “Hospitals are more full than they’ve been throughout the COVID pandemic,” with more than 80% of beds occupied nationwide, and hospitalization levels continue to rise, especially for seniors. Flu levels are also increasing, and COVID levels are on the way up, too. Health officials in major cities are urging people to mask in public indoor spaces.
Commentary: It ain’t just COVID anymore, folks. It may also get worse before it gets better. We are thankful that there are no more stay-at-home orders in our future. However, that leaves only one option to take back control from the virus: prevention. How?
Wear a mask and wash your hands. Deciding not to wear a mask in public at this point because “the pandemic is over” is cutting off your nose to spite your face. Amazingly effective vs. both COVID and flu.
Test early and often. Tests are broadly available, and pricing is as low as it has been. Free tests are also still available for those on Medicare and private insurance, as well as via distribution programs in many states. Tests give you the power - use them and help make the best decisions for you and those around you.
Of course, get your vaccines and boosters - but you have heard that (from us and many others) before. It may not stop you from getting COVID, but it can keep you out of hospital, which is a very good thing.
Test to Treat - Expanded but not still not large enough?
There’s a lot to like about Test to Treat (TTT), the federal program that allows people to get diagnosed with COVID and receive treatment with Paxlovid during the same visit. But despite the administration's expansion of Test to Treat centers to 2,229 sites, a study by Brigham and Women’s Hospital and University of Virginia showed that 15% of the US population has to drive more than an hour to take advantage of it. No surprise that this challenge is most common in rural areas, where the percentage of people with hour-plus drives to TTT rises to 59%.
BQs in, Monoclonal Antibodies Out
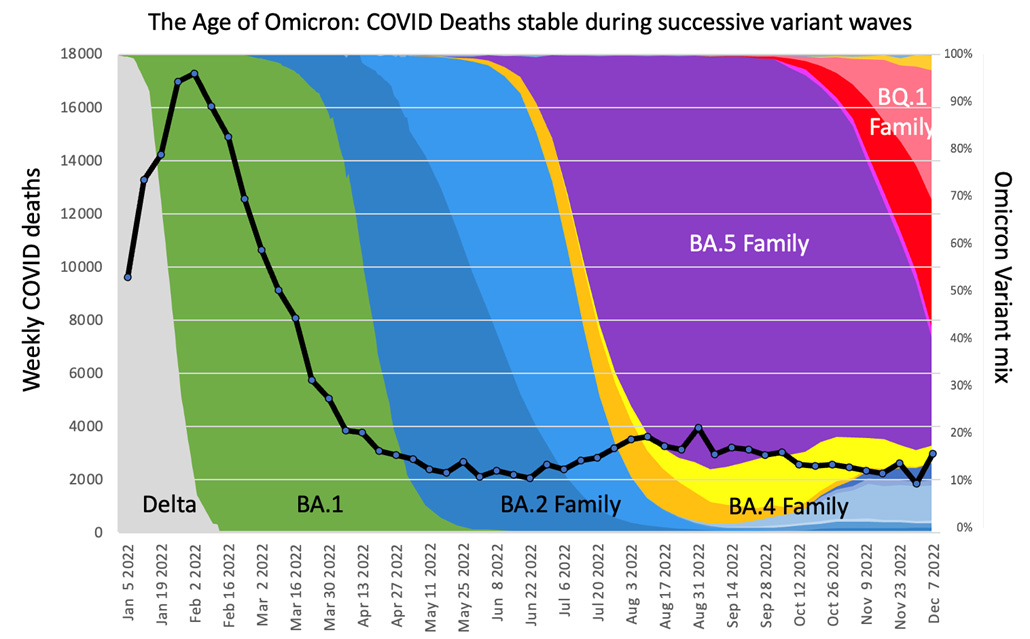
The update on the US variant mix: The BQ.1 family is now the dominant variant, at 68% of US cases. While the BQs aren’t causing a significant increase in the death rate as of yet (see chart below), that’s a lagging indicator. The big change right now is that this group, which is the most effective one yet at evading both vaccine- and infection-based immunity, has rendered the last remaining monoclonal antibody treatment (bebtelovimab) obsolete. The FDA has rescinded its EUA.
For now, Evusheld - the pre-exposure prophylaxis medication for immunocompromised folks - remains authorized and available. However, given that the drug combination is somewhere between 2000 and 10,000x less effective against the BQs than it was against the wild type, it seems unlikely that it will last a whole lot longer.
Food for Thought
Why is NGS so underutilized in cancer treatment?
It’s been nearly 25 years since the first molecularly targeted drug was approved by the FDA: Herceptin (trastuzumab) for metastatic HER2-overexpressing solid tumors in breast cancer. Gleevec (imatinib) for chronic myeloid leukemia (CML) followed in 2001, and since then FDA has approved over 90 targeted therapies: 27 molecular biomarkers for 40 drugs across 16 tumor types (see this 2019 review and a NEJM perspective this week). Beyond this, off-label use is commonplace, and best practice includes next-generation sequencing (NGS) for all tumors.
Despite that, NGS molecular profiling is highly underutilized across the vast majority of cancer types and patients. For example, a report at this year’s meeting of the American Society of Clinical Oncology stated that in non-small-cell lung cancer, profiling was performed for only 22% of the 37,925 patients for whom this would have aided therapy selection.
Tumor profiling has come a long way beyond the “I have a drug, find me patients'' companion-diagnostics era. Physicians now have a broad armory of effective therapies for particular driver mutations across many organ systems – therapies that block out-of-control growth signals and/or release suppressed regulatory pathways. While single-mutation therapies have very different effectiveness profiles across organ systems, and “cocktails” of these drugs have generally led to only temporary remission, enormous progress in immunotherapy offers more pan-tumor type effectiveness (e.g. checkpoint inhibitors PD-L1/PD1 and CAR-T). A 2018 study showed that 30% of 11,348 patients across seven major tumor/organ types could benefit from these therapies, based on NGS to assess the impact of microsatellite instability (MSI), high tumor mutational burden (TMB) and/or PD-L1 over-expression in their tumors (annotated Figure 2A shown here).
Commentary: Why is molecular profiling so underutilized? One underlying cause: the speed of progress in recent years has outpaced physician training and patient awareness. In a study released last week, oncologists cited lack of clinical relevance and/or inadequate evidence of clinical utility as the two most important reasons for not ordering NGS molecular profiling, but this is simply unwarranted today. Yes, this profiling may simply confirm that the planned therapy for some (perhaps even most) patients is the right one. For others, NGS may indicate that standard treatments are not likely to work, and that clinical trials may be the only realistic opportunity. But a substantial proportion of profiles will guide clinicians toward better-informed therapy selection that will give their patients additional months or years of healthy life. In all cases, the goal is the same: to give patients confidence that they are receiving the best treatment that 25 years of diagnostic progress has enabled.
Quick Hits
The FDA debuted a new step-by-step guide to help folks know when they need to test, how best to use OTC tests, and how to interpret the results. Our quick comment: The page isn’t mobile-friendly, and we still think asking folks without symptoms to take three tests in a row is a non-starter. But otherwise this guide is a great overview of everything most people need to know about when and how to use home tests.
The FDA has authorized the use of both the Pfizer and Moderna bivalent boosters in children as young as 6 months. As usual, nothing is simple. Littles up to age 5 who’ve had Moderna can be boosted with Moderna. Kiddos up to age 4 who haven’t finished their Pfizer primary series will now end the three-shot series with a Pfizer bivalent booster. But if kids in that age bracket are done with a primary series of Pfizer monovalent vaccines, they can’t get a bivalent booster yet - FDA is waiting on data that’s scheduled to arrive in January.







Education is key, but another critical reason lies in the challenges of reimbursement for NGS in oncology. With more consistent / feasible levels of NGS testing reimbursement for necessary biomarkers, clinical utilization of NGS will absolutely increase and drive more localized testing as hospitals / practice groups gain a better line-of-sight on payback for NGS technology investments. Currently, a majority of US oncologists / hematologists are reliant on outsourced large central labs, who they themselves, are struggling to remain solvent without external investments.