ALSO IN THIS ISSUE
New CDC COVID Dashboard: Genomic Surveillance of International Travelers
YLE’s Predictions for Fall Viruses
POC Test to Distinguish Between Viral and Bacterial Respiratory Infection
New and Noteworthy
Genomic Surveillance of International Travelers gets a Dashboard
The CDC’s COVID genomic surveillance of international travelers has expanded to four major airports (JFK, Newark Liberty, San Francisco, and Atlanta’s Hartsfield-Jackson). Results, which come from over 1,000 volunteers per week, now appear weekly on a new dashboard and include test positivity rates from pooled samples as well as which variants have been detected. (H/t to COVID-19 Data Dispatch for the heads-up.) Commentary: It’s a teeny, non-random sample of what COVID flavors international travelers are bringing into the US, but it’s better than nothing. And we get charts.
Your Epidemic Future is . . . Hazy
Hot off the presses is Your Local Epidemiologist Dr. Katelyn Jetelina’s assessment of the triple threat of flu, RSV and COVID for this year. Bottom line: good news on COVID, with declines in Singapore and Germany (the canaries in the coalmine for two scary novel variants XBB and BQ.1); mixed news on flu, which is off to a stronger-than-usual start here in the US, but for which the year’s vaccine is a good match; and bad news on RSV - growing quickly, especially in children. How this mix will pan out over the course of the fall and winter is anybody’s guess, but the combination is very worrisome. Commentary: Get vaccinated - for both flu and COVID. Our health care workers need all the help they can get.
Respiratory Virus or Bacterial Infection? POC Test Aims to Tell You - Quickly
In an ideal world, clinicians would be able to rule out viral causes of acute respiratory infections before prescribing antibiotics. Researchers in Massachusetts studied a 10-minute test that aims to do that by combining a myxovirus resistance protein A antibody (a marker of viral infection) with a C-reactive protein antibody (a marker of bacterial infection) in a simple one step lateral flow fingerstick cassette. In the study, inappropriate prescribing of antibiotics for viral infections was reduced by 37%; while failure to prescribe antibiotics for a bacterial infection was reduced by 82%. Commentary: While promising, results aren’t a slam-dunk yet. The most effective use case for the test is ruling out a bacterial infection rather than the converse (negative predictive value of 98.7% for bacterial infections). Still a very useful step in the right direction - we’re looking forward to more progress in this area.
Food for Thought
Crispr in Diagnostics will be a Revolution . . . Eventually.
Two years ago this month, the Nobel Prize in Chemistry was awarded to two of the pioneers of the Crispr DNA-editing technique. Since then it has displaced all other laboratory techniques for that purpose by virtue of its accuracy, simplicity, ease of use, and low cost.
How it Works and Why it was Nobel-worthy
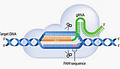
To use Crispr, you start by creating a target/guide string of ~20 RNA bases that are complementary to the location where you want to delete or modify a gene. You combine that with an enzyme (the original enzyme used in Crispr is an endonuclease called Cas9), mix the two with DNA, and the Crispr/Cas9 complex will bind to that exact location and edit it for you. (Of course, in practice it is a little more complex, but not much).
Until surprisingly recently, gene “editing” was accomplished by using radiation or chemicals to randomly mutate DNA (e.g., in thousands of fruit flies), then looking at all of the results to see what happened (red or white fruit fly eyes, anyone?). More recently, improved but complex mutation tools were developed, such as PCR-directed mutation and enzymatic techniques (Zinc Finger Nucleases - ZFNs or TALENS). All of these have been entirely obsoleted by Crispr.
How Crispr Diagnostics Work
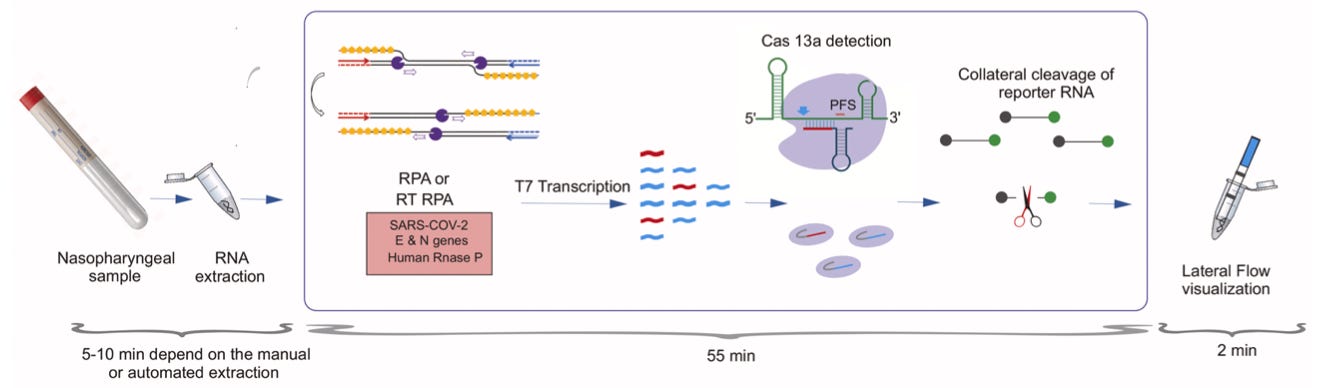
The pandemic accelerated research into the diagnostic uses of Crispr dramatically. Although just three Crispr-based SARS-CoV-2 tests are authorized (two versions of DETECTR from Mammoth Biosciences, plus one from Sherlock) a recent review summarized 36 novel Crispr techniques in development for diagnostics - with names like IMPACT, CASMEAN, CASCADE, SHINE, and SATORI. Combining Crispr with the enormous progress in microfluidics and lab-on-a-chip offers an exciting path forward.
A Crispr complex will only attach if and when it finds the unique stretch of the viral genome to which it was targeted (works for bacteria, amoeba, and fungi genomes, too), which is analogous to what PCR primers do. But PCR then does something Crispr cannot: once it finds its target, it generates billions of copies, the presence of which is easy to detect. Many copies means the sample is positive; very few copies means negative.
The PCR diagnostic process requires six basic steps: collect/neutralize sample; purify/extract RNA; convert to DNA; add primers to amplify the target when present; add generic fluorescent probes to identify amplified copies; report result. Crispr diagnostics basically require all these steps plus one: the addition of Crispr towards the end.
Crispr fluorescent probes are not generic - they only report when the Crispr/Cas complex binds to its correct targets - but if you have to do PCR anyway, is it worth the bother to do Crispr as well? As always, the devil is in the details, but one thing Crispr diagnostics do provide is double protection against some common PCR errors. If PCR amplifies the wrong target, Crispr will not be fooled.
All New Diagnostics Need Fine-Tuning, Crispr Not Excepted.
Forty years before Crispr, chemistry Nobel winner (and surfer) Kary Mullis sketched the concept of PCR on a napkin, but until a heat-tolerant polymerase was identified from a Yellowstone hot spring, PCR was labor-intensive and slow. Crispr diagnostics have just begun this fine-tuning process, 38 years behind PCR, but enormous progress is being made.
One example of this progress is the discovery of novel Cas enzymes which expand on Cas9’s properties. Cas12a (Mammoth) and Cas13a (Sherlock, illustrated below) find target DNA the way Cas9 does, but then they also cut any nearby single strand DNA (ssDNA). If you include short ssDNA strands attached to fluorescent reporter molecules, it is easy to see when binding has occurred. (Cas12a binds DNA, while Cas13a binds RNA directly without it being converted to DNA first.)
Nevertheless, Crispr’s need for prior amplification (e.g. PCR or isothermal) remains, and it’s the single biggest challenge for the technique’s use in diagnostics. Many clever ideas to deal with the problem have been floated, but none have yet overcome (or minimized) this disadvantage versus PCR.
EUA Update
The FDA issued one new EUA, 13 amendments to existing EUAs, and three safety communications (two EUA Revocations and one Warning Letter) and one marketing clearance in the last four weeks (since the September Newsletter).
New EUAs (1):
Molecular (1): Metrix Covid-19 Test (Home Saliva Kit)
Amendments to Existing EUA’s (13):
Molecular (7): Quidel Lyra | Abbott ID NOW | Quest PCR DTC | Cepheid Xpert Xpress | GeneMatrix NeoPlex | Luminex NxTAG | Cleveland Clinic SelfCheck cobas
Antigen (6): InBios Int’l | Siemens CLINITEST | SD Biosensor Pilot Home Test | Xiamen Boson | Access Bio CareStart | iHealth Labs
Safety Communications (3):
Revocations (2): Cleveland Clinic SelfCheck COVID-19 TaqPath Multiplex PCR | Cleveland Clinic SARS-CoV-2 Assay
Warning Letters (1) Lakpura LLC
510(k) clearances (1):
Roche’s PCR test. This test is the second to move from EUA to the 510(k) designation, after BioFire’s Respiratory Panel 2.1, a multi-analyte diagnostic, was granted marketing authorization using the De Novo review pathway in March 2021.
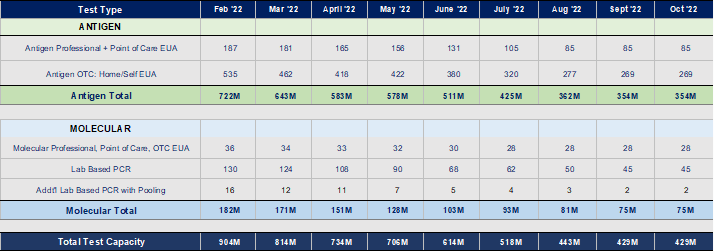
Monthly Capacity: Current EUAs
May June July Aug Sept Oct
706 614 518 443 429 429
No changes in capacity estimates this month. Manufacturers and labs are broadly holding capacity steady for the possibility of surge(s) through the winter.
Quick Hits
Results from a small study published in JAMA Network once again indicated that five days of isolation for COVID simply isn’t long enough. In the cohort of 268 patients studied, 27% of subjects were still testing positive seven days after their initial positive test.
The White House released a new National Biodefense Strategy and Implementation plan. More to come, including our analysis, next week.